
Research Interests
Regulation of Gene Expression, Protein-DNA Interactions, Assembly of the Transcription Apparatus
Research in Dr. Stumph's lab is aimed at understanding the molecular mechanisms of gene expression in higher organisms. Dr. Stumph's group has been characterizing and studying the expression of genes that code for the small nuclear RNAs (snRNAs) known as U1, U2, U4, U5, and U6. The snRNAs are a special class of RNA molecules that are involved in messenger RNA (mRNA) splicing (the removal of intron sequences from mRNA precursors). The genes coding for the U-snRNAs are interesting to study not only because they code for RNA molecules with such fundamental importance to cellular metabolism, but also because their expression appears to be controlled by unique mechanisms. For example, most snRNA genes are transcribed by RNA polymerase II (the same enzyme that synthesizes mRNAs), but these Pol II-transcribed snRNA genes contain no introns and they lack the TATA sequences commonly found upstream of mRNA transcription units. In contrast, U6 snRNA genes contain TATA boxes but are transcribed by RNA polymerase III.
Despite this difference in RNA polymerase specificity, the promoter sequences of U6 genes are actually very similar to the promoters of U1-U5 genes. In the fruit fly Drosophila melanogaster, a 21-base-pair-long cis-acting sequence, termed the proximal sequence element, or PSEA, is uniquely required for the transcription of both classes of snRNA genes. The PSEA specifically interacts with a multi-subunit protein termed the small nuclear RNA activating protein complex, DmSNAPc. DmSNAPc is required for the transcription of U1 and U6 snRNA genes by RNA polymerases II and III respectively. Our goal is to understand how the same protein can in one case recruit RNA polymerase II (for U1 transcription) but in another case recruit RNA polymerase III (for U6 transcription). In order to answer this question, we apply various biochemical and molecular biological techniques to the study of the Drosophila system.
The U1 and U6 PSEA sequences are identical at 16 of the 21 positions. However, we have shown that the 5 nucleotide differences between the U1 and U6 PSEAs are sufficient to determine the RNA polymerase specificity of the U1 and U6 gene promoters. Furthermore, by site-specific protein-DNA photocrosslinking techniques, we have determined that DmSNAPc consists of at least three distinct polypeptide chains that contact the DNA of the PSEA. Significantly, the data also reveal that the precise contacts made between the protein and the DNA are different depending upon whether DmSNAPc is bound to a U1 or U6 PSEA sequence.
From these data, we have proposed that the U1 and U6 PSEA sequences act as differential allosteric effectors of DmSNAPc. According to this model, when DmSNAPc binds to a U1 PSEA sequence, it adopts a conformation that allows it to recruit only RNA polymerase II basal factors during subsequent steps of pre-initiation complex assembly. On the other hand, when DmSNAPc binds to a U6 PSEA, it adopts a conformation compatible with the recruitment of only RNA polymerase III basal factors. This is illustrated schematically in the figure below.
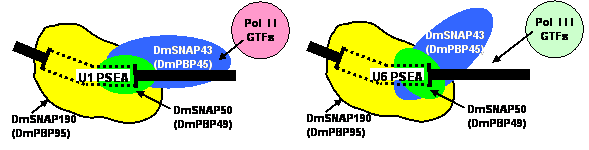
DmSNAPc binds to U6 PSEA
We are currently making mutations in these various subunits to identify domains within the proteins that are required for complex assembly, DNA binding, and the activation of transcription. One goal is to identify mutations that will result in the loss of activation of transcription by one RNA polymerase but not the other. In this way we hope to identify domains within the proteins that are specifically required for transcription by one RNA polymerase but not the other.
Recent Publications
- "Bdp1 interacts with SNAPc bound to a U6, but not U1, snRNA gene promoter element to establish a stable protein-DNA complex,"
Verma, Neha; Hurlburt, Ann Marie; Wolfe, Angela; et al.,
FEBS Letters 592, 2489-2498. (2018). (doi: 10.1002/1873-3468.13169.) - "TFIIIB subunit locations on U6 gene promoter DNA mapped by site-specific protein-DNA photo-cross-linking,"
Jin Joo Kang, Yoon Soon Kang, and William E. Stumph,
FEBS Letters 590, 1488-1497 (2016). (doi:10.1002/1873-3468.12185.) - "The Myb domain of the largest subunit of SNAPc adopts different architectural configurations on U1 and U6 snRNA gene promoter sequences,"
Yoon Soon Kang, Michelle Kurano, and William E. Stumph,
Nucleic Acids Research 42, 12440-12454 (2014). (doi:10.1093/nar/gku905.) - "Differential Utilization of TATA Box-binding Protein (TBP) and TBP-related Factor 1 (TRF1) at Different Classes of RNA Polymerase III Promoters,"
Neha Verma, Ko-Hsuan Hung, Jin Joo Kang, Nermeen H. Barakat, and William E. Stumph,
Journal of Biological Chemistry 288, 27564-27570 (2013). (doi:10.1074/jbc.C113.503094.)
